ARCHIVES
Potential Briefs
ISSUE #05 — November 2011
Comment on this Newsletter... CLICK HERE
ANNOUNCEMENTS
Introducing Quantum Neuromonitoring & Dhiraj Jeyanandarajan, M.D.
 ORIMtec is pleased to announce the acquisition of its assets by Quantum Neuromonitoring Corporation. Dhiraj Jeyanandarajan, M.D. holds the office of Medical Director and will retain ORIMtec officers Daniel Slepian, PA, CNIM, Director of Clinical and Surgical Services; Mark Twede, R. EEG T., CNIM, Manager of Clinical and Surgical Services; and Richard Slepian, Director of Business Operations.
ORIMtec is pleased to announce the acquisition of its assets by Quantum Neuromonitoring Corporation. Dhiraj Jeyanandarajan, M.D. holds the office of Medical Director and will retain ORIMtec officers Daniel Slepian, PA, CNIM, Director of Clinical and Surgical Services; Mark Twede, R. EEG T., CNIM, Manager of Clinical and Surgical Services; and Richard Slepian, Director of Business Operations.
Dr. Jey is a Board Certified Neurologist has completed a fellowship in neurophysiology with an emphasis on intraoperative monitoring. As Medical Director, Dr. Jey will be responsible for the direct supervision and professional oversight of all neurotechnologists.
You can be assured that Quantum will carry on ORIMtec's traditions and philosophy: to provide the most comprehensive, technically advanced monitoring services and resources using customized products for the benefit of patients and in collaboration with surgeons. Our mission is to enhance IONM services through continual improvements to training and education and to develop support products including reference materials and an interactive communications platform for the benefit of the monitoring community. We embrace the possibilities that emerge in our line of vision, and we strive to build on that potential. An evolutionary blend of medicine, technology and compassion continuously augments our success.
ARTICLE HIGHLIGHT
Dhiraj Jeyanandarajan, M.D. - Board Certified Neurologist
Article: Can Triggered Electromyograph Thresholds Predict Safe Thoracic Screw Placement?
Authors:Barry L. Raynor, BA, Lawrence G. Lenke, MD, Yongjung Kim, MD, Darrell S. Hanson, MD, Tracy J. Wilson-Holden, MA, Keith H. Bridwell, MD, and Anne M. Padberg, MS
Journal:Spine 2002 Volume 27, Number 18, pp 2030-2035
Summary: This paper is a prospective clinical study of thoracic pedicle screw placement and monitoring with triggered electromyographic (tEMG) testing in order to evaluate the sensitivity of tEMG in the rectus abdominus muscles when evaluating thoracic level vertebrae for pedicle wall breach. The study involved 92 patients with a total of 677 thoracic screws from T6-T12. Conclusions were that tEMG responses recorded from the rectus abdominus muscles can be used to assess thoracic pedicle screw placement from T6-T12. Screws stimming above 6.0 mA are considered to be safe without medial wall defects. And an absolute value of 6.0 mA should be compared with the mean of all other thresholds in each patient to provide additional information regarding the likelihood of medical pedicle wall defect.
Comments: While this article is one of the older ones on tEMG threshold values, it was the first well designed study for thoracic level screws. One key point for thoracic and lumbar pedicle wall thresholds is that it is always recommended to compare with the mean of other thresholds for that patient. For example, a thoracic threshold of 7.0 mA would still be highly suspect for medial wall defect if all the other thoracic levels tested above 15mA. The absolute cutoffs should never be considered 'absolute' in these circumstances.
This transitions well into a more quantitative method to help factor in variables such as bone density into the equation when testing pedicle screws. My esteemed colleague, Dan Slepian, has long advocated testing bone thresholds in order to establish a baseline to which the absolute pedicle thresholds can be based off of. This would take into account the different variables inherent in each patient to give a more complete picture. This is an excellent topic for future study to help weigh the true value of that additional data weighed against the time required to obtain it, etc. Please refer to Dan Slepian's "Technical Review" (below) Bone Thresholds for Pedicle Screw Stimulation to learn more about this technique.
TECHNICAL REVIEW
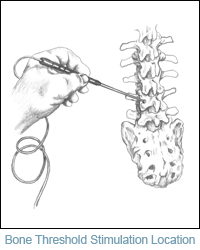
Bone Thresholds for Pedicle Screw Stimulation
Daniel Slepian, PA, CNIM
While numerous articles have been published pertaining to pedicle screw stimulation, few articles mention the need for establishing a bone density threshold prior to inserting the implant. This article explores the need to establish a bone threshold prior to screw insertion to minimize false positive findings. Additionally, little is said about the patient's age or the relative size of the pedicle in the three primary regions of the spine. Optimal bone thresholds should be set with consideration given to the patient's age, overall health and the differing sizes of the pedicle. If neurotechnologist's (NTs) are to use a specific stimulus intensity cutoff, such as 10mA to base a call on, they could be ignoring inherent physiological factors relative to optimal pedicle size and bone density.
Convincing the surgeon to establish a bone threshold prior to inserting a pedicle screw has not been readily accepted. As a result, NT's have found it was easier to convince the surgeon to establish a bone threshold when the screw tests positive at intensities below 10mA. Another point to consider is that the bone threshold may not be the same at any one level when compared to the opposite side. To date a bone threshold is obtained when a screw tests positive for triggered electromyographic activity. Based on this threshold NT's can confidently advise the surgeon of whether or not the implant has in fact breached the medial wall of the pedicle. Additionally, the technologist should always suggest that the surgeon obtain an image for verification at this point in the surgery.
Surgical data on common bone threshold levels in the lumbar spine has shown intensities in excess of 45mA. It is of no value to use intensities this high and this may in fact cause injury to adjacent nerves or the spinal cord. We recommend intensities not to exceed 30mA in the lumbar spine; 25mA in the lower thoracic spine, T6-T12; 20mA in the upper thoracic spine; and no higher than 15mA in the cervical spine. A significant amount of data is still needed at all levels... MORE
NEUROTECHNOLOGIST REFERENCE GUIDE
Third Edition
 You have your mind-set and equipment prepared for your 7:30 a.m. anterior cervical fusion. As you travel to the OR suite, you anticipate the modality sequence. The evoked potentials and EMGs to be monitored and electrode selection are ready, and so are you.
You have your mind-set and equipment prepared for your 7:30 a.m. anterior cervical fusion. As you travel to the OR suite, you anticipate the modality sequence. The evoked potentials and EMGs to be monitored and electrode selection are ready, and so are you.
When you check in with the charge nurse to verify your case, she says, "I have an anterior/posterior thoracolumbar procedure scheduled with the surgeon, not an ACF." A look of surprise crosses your face and you inquire again to verify the procedure. The charge nurse points to the schedule, looks up at you, shrugs her shoulders, and says, "Sorry, a thoracolumbar is scheduled." This isn't the first time a switch like this has happened and most likely will not be the last. It's a simple matter to deal with because you are well-versed in changing directions in midstream.
You take out your ORIMtec Neurotechnologist Reference Guide and simply refer to section 4's procedure-specific modality, turning to "Thoracic Procedures T2–T12." After reviewing the suggested modalities, you quickly shift your thought processes. Once your laptop fires up, you enter the new protocol. After referring to the guide again, you select the muscle groups and identify the needle insertion points. You are back on track and ready to go when the surgeon enters the OR 5 minutes early. This scenario highlights the value of using the Quanmtum Neuromonitoring Neurotechnologist Reference Guide as an adjunct to your existing skills, training, and experience.


